Off-label drug use
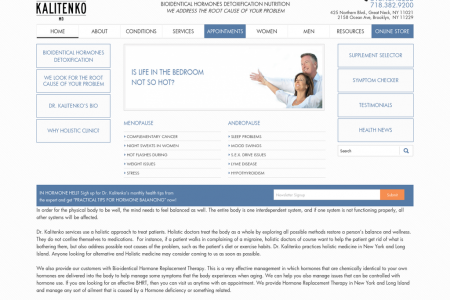
OFF-LABEL DRUG USE HAPPENS, IS SIGNIFICANT, IS DRIVEN BY RESEARCH, 41-58%metoprolol off label 100% 90% other off-label 80% 70% not coded or unclear 60% 50% other heart disease (off-label) 40% heart failure (off-label until 2001) chest pain (approved) 30% heart attack (approved) 20% 10% high blood pressure (approved) 0% 1999-2001: Cases of metoprolol pre- scriptions for heart failure (no. 1 killer of adults in US) jump by over 250% compared to 1998. 1999: Research 1992: Released & FDA-approved for high suggests effective- blood pressure, heart attack and chest pain 2001: FDA approves for heart failure. Prescriptions for heart failure increase ness for heart fail- ure 8% over the next year. US and FOREIGN APPROVALS, WORD-OF-MOUTH, 32-69%cymbalta off label 100% other off-label 90% not coded or unclear 80% 70% 60% chronic pain (off-label until 2010) 50% 40% 30% psychiatric (approved) 20% 10% 2004 2005 2006 2007 2008 2009 2010: FDA approves 2007: Canada ap- proves for treatment of chronic pain 2004: Released & FDA-approved for depression 2005: Some doctors suspect effective- ness in treating pain. Prescriptions for chronic pain increase 17-fold over 2004. for treatment of chronic pain IS RISKY & DANGEROUS, BUT ALSO SAVES LIVES. 57-95%methadone off label/debated 100% other off-label 90% not coded or unclear 80% trauma (off-label) 70% 60% 50% 40% non-cancer pain (debated; mostly off- label/inappropriate*) 30% 20% cancer 10% addiction (approved) (approved) 0% 2009 * Morbidity & Mortality Weekly Report. Vol 61. July 3rd, 2012. Centers for Disease Control and Prevention 1947: FDA approves for treating substance abuse & pain Background: In the US, doctors prescribe drugs to treat any condition they see fit, even if it is not ap- proved by the Food and Drug Administration (FDA) for that condition. These uses are considered “off- label" and make up over 20% of all prescriptions in the US. We collected over 3.7 million records of patient prescription data and associated medical conditions from 14 different Medical Expenditure Panel Surveys* over the period from 1996-2009. We processed, aggregated and linked this data across multiple years, patients and drug name equivalents, applying textual analysis and other filtering methods as appropriate, to obtain a sample large enough to analyze for off-label drug use by three-digit and top-level ICD-9 medical condition codes. The percentages provided reflect the minimum and maximum share of surveyed prescription cases within a span of one year that were for off-label conditions. The three drug-use timelines highlighted here were chosen based on recent publications and interest for medical professionals. However, our aggre- gated dataset provides easy access to analagous analyses by condition codes for over 2,000 medications. To make the above visualizations accessible to the lay person, we coded the reported three-digit ICD-9 condition codes for Metoprolol, Cymbalta and Methadone into approved and off-label subcategories with the help of a medical professional. The Los Angeles Times, Forbes Magazine, the Veterans Administration, physicians writing to the New England Journal of Medicine, and many other medical organizations have each made calls for better measurement and understanding of off-label drug use in the last year. Each of these calls comes as a direct result of reported increases in morbidity and mortality from non-approved uses of drugs in the US. Our work provides the first library we are aware of with estimates of off-label drug use in the US for a large number of (ie. greater than 10) medications. * available at http://www.healthdata.gov/ 9661 Z66I 1998 1997 . 6661 0007 2001 8661 2002 666I 2003 100z 2003 0007 2004 2005 2004 9007 ZO07 2008 2005 6007 LO07 2008 9007
Off-label drug use
Source
http://healthdata.govCategory
HealthGet a Quote